- A. Melts that erupt at the surface of the Earth and cool are volcanic. Magma that erupts to the Earth's surface is called lava.
- B. Melts that cool beneath the Earth are called plutonic.
- A. Texture
generally describes the grain size (or crystal size) in rocks.
- 1. A large or coarse grain size (> 1 mm) is associated with plutonic, or intrusive rocks. Slow cooling usually causes this texture.
- 2. Small or fine grain size (< 1 mm) is associated with volcanic, or extrusive rocks. Rapid cooling usually causes this texture.
- 3. Sometimes magma cools so quickly that crystals have no time to form. Instead glass forms (example, obsidian).
- B.
Composition of igneous rocks reflects the chemistry of the original
magma.
- 1. Mafic rocks are richer in Mg, Fe, and Ca. They are also darker in color and denser.
- 2. Felsic rocks are richer in K, Na, Al and Si, and, compared to mafic rocks, are lighter in color as well as density.
- 3. Intermediate compositions lie between these two end members.
- C. When taken together, each composition with each texture has a unique rock name. See the table below.
| Felsic | Intermediate | Mafic | |
|---|---|---|---|
| Coarse- grained |
granite | diorite | gabbro |
| Fine- grained |
rhyolite | andesite | basalt |
| Minerals | quartz, K-feldspar, amphibole, muscovite, biotite | plagioclase, quartz, amphibole | olivine, pyroxene, Ca-feldspar (plagioclase) |
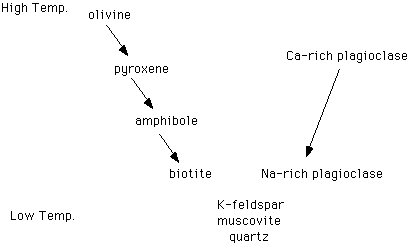
II. Igneous rocks are composed of silicate minerals, as discussed in the previous lecture. The relationship between these minerals in igneous rocks is described in Bowen's Reaction Series. See below:
|
- A. The
discontinuous series, on the left side of the chart, shows how one
silicate mineral crystallizes with respect to another at different
temperatures.
- 1. Minerals with simpler silicate crystallography occur at higher temperatures. More complex silicate crystallography forms at lower temperatures.
- 2. The temperature variation in Bowen's Reaction Series is roughly 700°C to 1100°C.
- B. The
continuous series, on the right side of the chart, shows how Ca and Na
vary with temperature in plagioclase.
- 1. More calcium-rich plagioclase forms at higher temperatures; more sodic plagioclase forms at lower temperatures.
- 2. The continuous series is an exception to the trend found in the discontinuous series. Plagioclase has a complex crystal structure, in which each silicate tetrahedron shares four of its oxygens with other tetrahedra. Similarly complex silicate minerals typically form at lower temperatures. Calcic plagioclase, however, forms at high temperatures.
- C.
Bowen's Reaction Series relates igneous mineralogy and crystallization
temperature to different magma types.
- 1.
Mafic magmas tend to have olivine, pyroxene, and calcic plagioclase.
- a. Mafic rocks tend to crystallize at higher temperatures
- b. Mafic magmas also tend to be less viscous (that is, they are runnier) than felsic magmas. This is related to the formation of minerals with simpler crystallography (silica tetrahedra are less likely to share oxygens with other tetrahedra).
- 2.
Felsic magmas tend to have amphibole, micas (biotite, musovite),
quartz, and Na- and K-feldspars.
- a. Felsic rocks usually crystallize at lower temperatures.
- b. Felsic magmas also tend to be more viscous (that is, gooey, like cold molasses). This is related to the formation of minerals with more complex crystallography.(silica tetrahedra are more likely to share oxygens with other silica tetrahedra).
- 1.
Mafic magmas tend to have olivine, pyroxene, and calcic plagioclase.